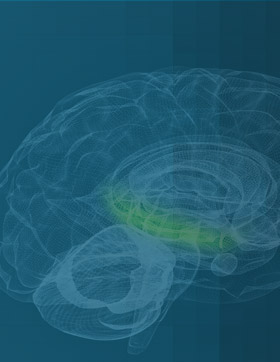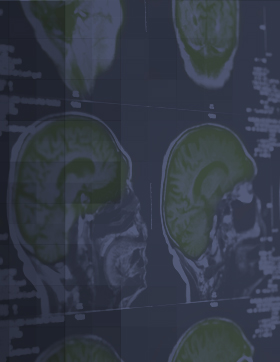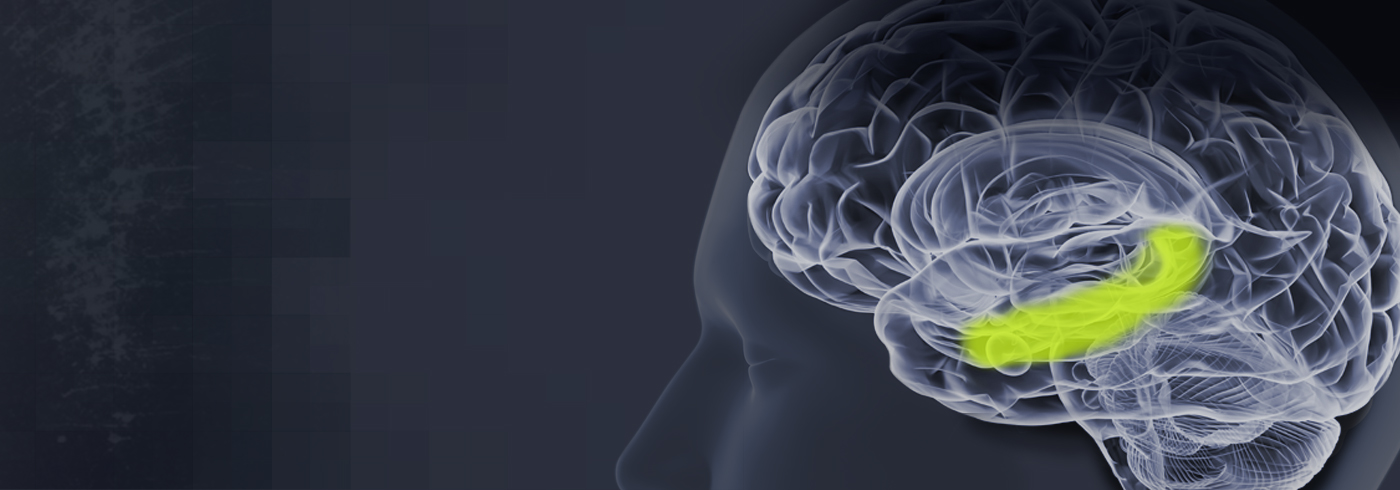
AGB101 is in development to treat mild cognitive impairment due to Alzheimer’s disease (MCI due to AD) and to control progression to Alzheimer’s dementia.
AGB101 is being investigated for the treatment of hippocampal overactivity and to assess its ability to control progression to Alzheimer’s dementia. Currently there are no treatments that have shown these benefits.
- AGB101 is being studied in patients with mild cognitive impairment due to Alzheimer’s disease (MCI due to AD), the earliest symptomatic stage characterized by memory loss
- No approved treatments for the 5.6 million US MCI due to AD patients (25 million worldwide)
- First and only product candidate being evaluated to treat the condition of hippocampal hyperactivity that leads to memory loss and brain atrophy
- Hope4MCI (Phase 2b trial) data acquisition was completed November 2, 2022 (last patient visit). The database lock and unblinding will now occur Q4 2022, with data analysis forthcoming in Q1 2023.
Learn more about the HOPE4MCI trial and clinical therapeutic program
AGB101 Is Being Evaluated to Determine if it treats symptoms and controls further progression
Our AGB101 Development Program
Intervening Early in the Disease Process
AGB101 is being studied for MCI due to AD, the symptomatic, pre-dementia stage of Alzheimer’s. MCI represents a period of initiation of marked changes in both brain function and memory, providing the best stage of the disease in which to measure efficacy. Currently FDA-approved treatments are indicated to provide symptomatic relief much later in the disease process when there is substantial neuronal cell death.
Known Molecule Being Studied at a Lower Dose
AGB101 is a proprietary extended-release formulation of levetiracetam, an FDA-approved anti-epileptic. AGB101 is being evaluated for MCI due to AD as a once-daily dose that is given to patients at approximately one-twelfth of the dose most commonly prescribed for epilepsy.
AGB101 is not approved by the FDA. The safety and efficacy has not been established.